Proteomic Analysis of Chromatin
PhD Position - 2025 IMPRS-EBM Recruitment
For understanding chromatin regulation in signaling, development and disease, it is important to elucidate changes in both histone posttranslational modifications (PTMs) and the composition of chromatin regulatory (multi)protein complexes.
Goal
We are concerned with proteomics method development in order to improve (sensitivity, quantitative accuracy) the discovery of novel chromatin and transcriptional regulatory mechanisms.
Approach and current efforts
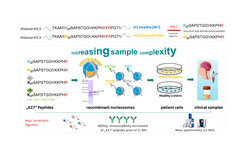
To pursue this we apply state-of-the art sample preparation methods (SP3/PAC, iST, C18-STAGE tipping) and LC-MS measurement approaches (quadrupole-orbitrap: DDA, DIA, PRM; timsTOF HT: DDA-PASEF, DIA-PASEF,). In our newest line of research that is funded by the BMBF Clusters4Future program (nanodiag BW), we are developing an immunoaffinity mass spectrometry approach (AffiPep), which aims to enrich a subset of histone PTM peptides prior to LC-MS analysis. In this context, we are especially interested in the histone H3 K27 oncohistone (K27M) mutation (figure: upper panel) that leads to the development of brain tumors (glioma) as well as other prominent H3 K27 modifications (K27methylation, K27acetylation). Starting with “spike-in” “K27” peptides and ArgC proteolytic digestion of histones, we will challenge the workflow by increasing the sample complexity with the ultimate goal of measuring (identification and quantification of “K27” peptides) patient samples by both LC-MS and biological nanopore sequencing (figure: lower panel). Overall AffiPep selection should ease the robustness, sensitivity and reproducibility of interrogating changes in the histone modification landscape of precious samples.