Novel tools for understanding immune cell movement in tissues
Lämmermann Lab
Project
In adult tissues, immune cells encounter a broad range of ECMs, including BMs and collagen-rich interstitial matrices, which vary largely in the specific biochemical composition and topology. Over the last two decades, it was realized that the mechanics of immune cells (but also other cells, e.g. cancer cells) in physiological, three-dimensional (3D) tissue environments can fundamentally differ from the migration strategies used by cells on two-dimensional (2D) surfaces (Lämmermann and Sixt, 2009; Lämmermann and Germain, 2014). Thus, analyses in native 3D-matrices are indispensable for migration studies to reflect influences by physical properties and geometry of the extracellular surrounding, in particular the ECM. For example, immune cells can switch to an integrin-independent mode of migration in 3D matrices and confined environments (Lämmermann et al., 2008). Thus, there is a requirement to study cell migration within the physiological context of tissues, where BMs, interstitial matrices and interspersed cells form 1D fibers, 2D surfaces, confined spaces, fibrillar 3D networks, elastic and stiff substrates in close proximity. Still, it is poorly understood how all these variables are integrated to shape the behaviour of migratory cell types and in reverse, how cells shape their environment during migration.
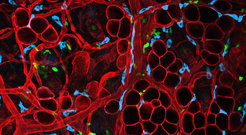
Even by the means of state-of-the-art two-photon intravital microscopy, the correlation of migrating cells to tissue geometry has been limited to the visualization of the collagenous fiber network (2nd harmonic generation), elastin fiber network (3rd harmonic generation) or stromal cells. However, we are still lacking fundamental knowledge on how other ECM components are either modified during migration, or how they contribute to the migration modes and patterns of migrating cells. To overcome current experimental limitations to directly visualize ECM components during cell migration in tissues, we collaborate with the research group of Sebastian Arnold (University of Freiburg) as part of the SFB850 to develop, characterize and apply novel tools and approaches for the visualization of migratory cell behaviour and cell-ECM interactions in physiological 3D tissue environments.
