Stem cell pluripotency and higher-order chromatin structure
We found that the nuclear proteins Satb1 and Satb2, which function as determinants of higher-order chromatin structure, have opposing roles in the regulation of the pluripotency gene Nanog. In particular, Satb1 represses Nanog, whereas the closely related Satb2 protein activates Nanog.
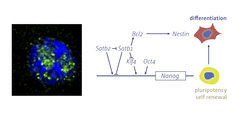
Moreover, both Satb1-deficient ES cells and wild-type ES cells in which Satb2 is overexpressed are more efficient in reprogramming human B lymphocytes in heterokaryon fusion experiments. Recently, we found that SUMOylation of Satb2 during ectodermal differentiation is required for the down-regulation of pluripotency genes. Satb2 also plays a role in B lymphocytes by binding to AT-rich sequences that flank the intrinsic immunoglobulin heavy chain (IgH) enhancer.
By studying the subnuclear localization and higher-order chromatin structure of the IgH locus in Satb2-deficient pro-B cells, we anticipate to unravel the molecular basis of Satb2 function in the regulation of higher-order chromatin structure. We combine biochemical, imaging and genetic approaches, to elucidate how Satb proteins functionally organize chromatin.
