How do chromosomes become organized de novo during early development?
Iovino Lab
Chromatin inside the nucleus is organized in complex hierarchical folding patterns, including chromatin loops, topologically associating domains (TADs) and compartmental domains. These fundamental features are de novo established after fertilization. Early embryogenesis, therefore, provides the opportunity to identify the molecular players that drive the establishment of the 3D genome structure. In mammals, CTCF and cohesin mediate the formation of loops and give rise to most TAD boundaries. Depletion of CTCF and cohesion causes TAD defects, but chromosome compartments and higher order chromatin organization are maintained, suggesting that other mechanisms are in place.
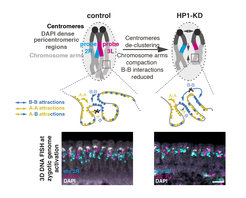
HP1a is an essential protein present throughout early embryogenesis, before the establishment of 3D compartmental domains, in vertebrates and invertebrates. HP1a is known to localize to repeat-rich, gene poor heterochromatin where it is likely to form phase-separated nuclear microcompartments.
We discovered a novel and fundamental role for HP1a in the reorganization of chromatin after fertilization. We show that HP1a i) controls the general folding of chromosome arms, ii) controls the clustering and compaction of heterochromatic pericentromeric regions and iii) contribute to the formation of the B compartment. Unexpectedly, we also discovered that the A compartment is generated through an HP1a-independent mechanism.
